CNA CONCENTRATED NITRIC ACID (Direct Process)
Overview
Concentrated nitric acid (CNA) is used in the production of explosives and chemicals such as isocyanates and nitrobenzene. Such concentrated nitric acid, is made thru a complex process explained below.

Nitric acid and water form an azeotropic mixture at 70% HNO3. Some processes use dehydrating chemicals such as sulphuric acid or magnesium nitrate to obtain concentrated acid by extractive distillation of the azeotrope at the expense of high consumption of steam. This CNA process integrates the production of weak and concentrated HNO3 and avoids the use of additional chemicals, that reduces the steam consumption.
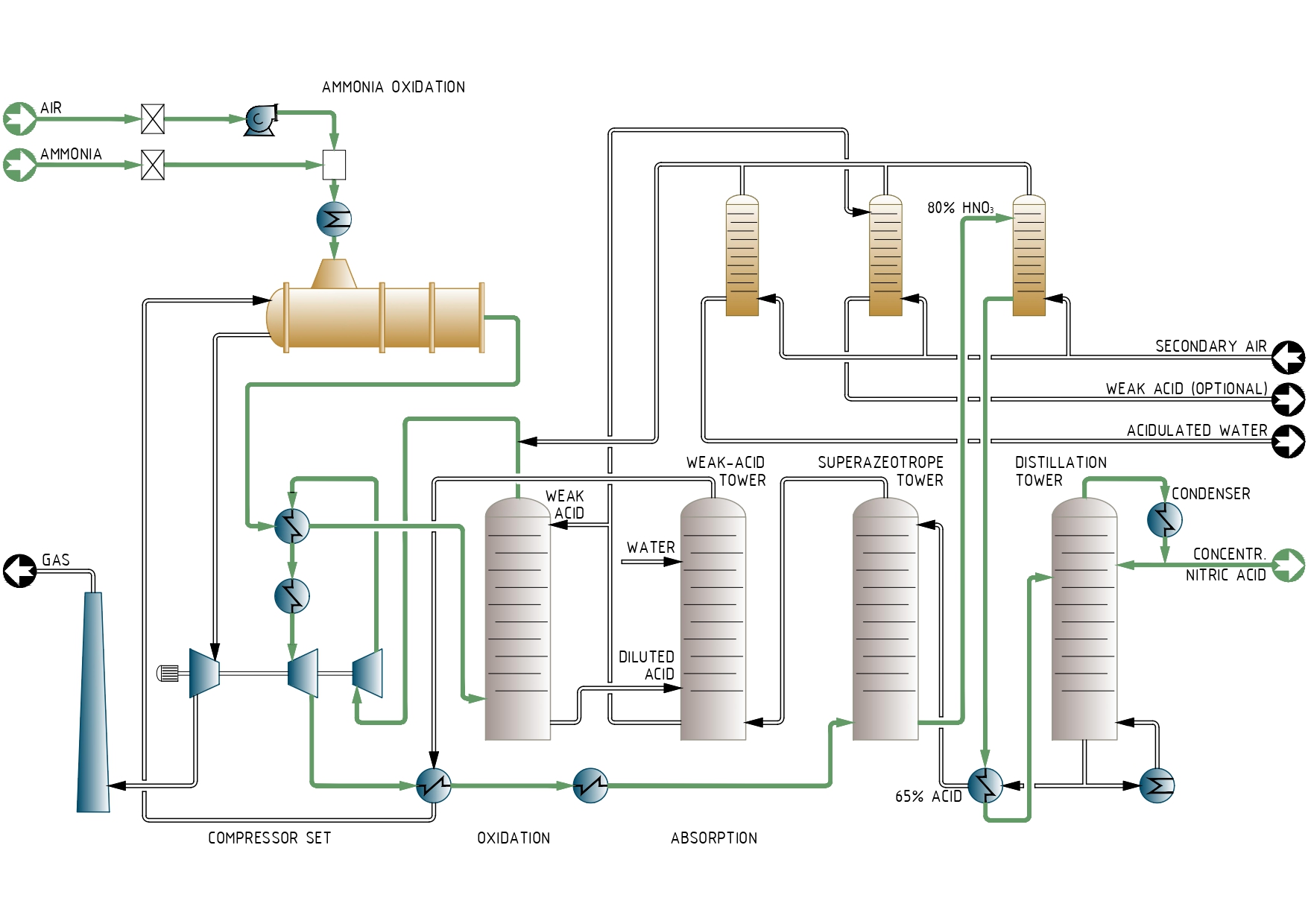
To manufacture CNA, ammonia is oxidized with air over a catalyst, producing NO that is oxidized to NO2 at later stage.
The nitrogen dioxide is then absorbed in water to make the acid. In ESPINDESA technology, gases after ammonia oxidation are enriched in NO2 and absorbed in azeotropic acid to produce a superazeotropic acid (80% HNO3) that is then distilled to CNA.
The process can simultaneously produce concentrated and/or weak acid: the product mix can be changed by adjusting the position of two valves. In addition, most of the towers and exchangers are made of conventional austenitic stainless steel.

Compared to other processes, this CNA route requires lower investment in plants sizing 100-250 metric ton/d than traditional extractive distillation.
Process Description
Liquid ammonia is evaporated and oxidized with air to form NO at atmospheric pressure and 810º- 830°C:
4 NH3 +5 O2 ==> 4 NO+6 H2O
The temperature of the process gas is lowered to its dewpoint in several heat exchangers, which preheats tail gas, produce steam and preheats boiler feedwater. Most of the water formed during reaction is conde3nsed in a cooler condenser as an acidic stream (below 1.0% w/w HNO3). This water is bleached with air to remove nitrogen oxides and eliminated from the plant to maintain the water balance.
The dry gas stream is preheated and sent to the bottom of the oxidation tower. Here, 60% acid enters the top of the sieve-trays column. As the gas and liquid flow countercurrently, the acid oxidizes NO to NO2:
NO+2 HNO3 ==> 3 NO2 + H2O
Three moles of NO2 are produced per mole of NO, givinga stream very rich in NO2 and N2O4 (formed by the dimerization of NO2). This reaction establish the necessary conditions to obtain superazeotropic acid: high partial pressure of nitrogen dioxide.
Diluted acid from the bottom of the oxidation unit is recycled into the absorption section for concentration. The process gas leaving the top is mixed with the secondary air containing nitrogen oxides and compressed to about 10 bar.
Absorption section
This section consists of two columns: one produces the superazeotrope, the other makes weak acid.
The gases first enter the superazeotropic column where NO2 is absorbed in azeotropic (70%) acid from the distillation column and 80% nitric acid is formed.
This superazeotropic acid saturated with nitrogen oxides, is bleached with air, preheated and then fed to distillation column where 70% azeotropic acid is obtained from the bottom and concentrated nitric acid, 99% concentration, is obtained from the top.
Weak acid
Since the process gases leaving the top of the superazeotropic absorption tower are still rich in nitrogen oxides, they are sent toa second absorption tower. Water is fed at the top, and low concentration acid from the oxidation tower enters in the intermediate trays. 60-65% acid is obtained from the bottom, and subsequently bleached with air.
The tailgas is preheated to about 400ºC and flows into the expander to recover about 50% of the energy required for compression. After expansion, tailgas is sent to the stack.
OPERATING REQUIREMENTS
(Per metric ton of HNO3)
Ammonia, 281 kg
Electricity, 350 kW/h (Compressor by elect. motor)
Steam import, 0 kg
Cooling Water, (T=l0°C) 125 m³
Catalyst, net (as Pt) 55 mg
Process Flow Diagram